 
It considers both bonding and lone electron pairs in determining molecular geometry. According to VSEPR theory, electron pairs surrounding a central atom will repel one another to achieve a spatial arrangement that minimizes repulsion and stabilizes the molecule. The VSEPR (Valence Shell Electron Pair Repulsion) theory is fundamental in predicting electron geometries.As a result, its electron geometry is determined to be tetrahedral, meaning the molecule adopts a shape resembling a pyramid with a triangular base. Importantly, methane lacks any lone pairs. 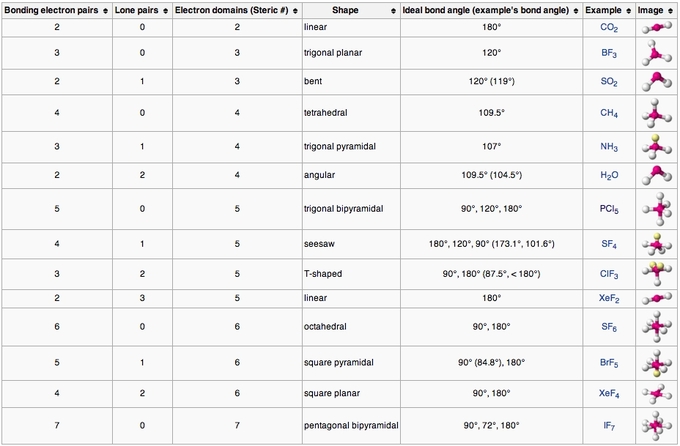
Therefore, there are a total of eight electrons around carbon. The four hydrogen (H) atoms share an electron pair with carbon, forming four covalent bonds. In methane, the central atom is carbon (C), which possesses four valence electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
